Selenium & Iodine: The Essential Duo for a Healthy Thyroid and Metabolism
In the vast, intricate symphony that is the human body, countless tiny players perform their roles, often unseen, yet absolutely critical to the grand orchestration of life. Among these microscopic maestros, two trace elements stand out for their profound and non-negotiable partnership in conducting one of the body’s most vital systems: the thyroid gland. These are Iodine and Selenium, an essential duo whose synergistic dance dictates not only thyroid health but, by extension, the very rhythm of our metabolism, energy levels, mood, and overall well-being.
To the knowledgeable observer, the relationship between these two elements is not merely additive; it is a story of codependence, protection, and activation – a biological ballet where the absence or imbalance of one can throw the entire performance into disarray. This article delves deep into their individual tales, their intertwined destiny, the mechanisms of their synergy, and the profound implications for human health when this delicate balance is disrupted.
Part 1: Iodine – The Architect of Thyroid Hormones
Our journey begins with Iodine, an element whose story is etched into the very fabric of human history, marked by both ancient suffering and modern triumph. Discovered in 1811 by Bernard Courtois, a French chemist, Iodine quickly revealed its unique affinity for the thyroid gland, a small, butterfly-shaped organ nestled at the base of the neck. This affinity is not coincidental; it is the cornerstone of thyroid hormone synthesis.
The Element's Tale: Iodine, atomic number 53, is a non-metallic element, a halogen, distinguished by its violet vapor. Its scarcity in many terrestrial environments, particularly inland regions away from the ocean, historically led to widespread deficiency. Before the advent of iodized salt, goiter – a grotesque enlargement of the thyroid gland – was a common sight, a visible manifestation of the body's desperate attempt to capture more of the scarce iodine needed for its vital function. More tragically, severe iodine deficiency during pregnancy led to cretinism, a devastating condition characterized by severe mental retardation and stunted physical growth, underscoring iodine’s critical role in neurological development.
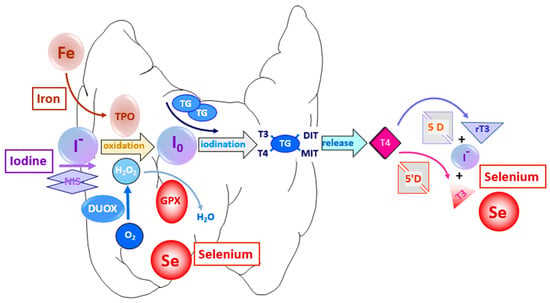
Thyroid's Affinity and the Master Builders: The thyroid gland possesses a remarkable biological pump, the Sodium-Iodide Symporter (NIS), which actively concentrates iodide (the ionic form of iodine) from the bloodstream, often to levels 20-50 times higher than in the plasma. This concentrated iodide is then transported into the thyroid follicular cells, where the true magic of hormone synthesis begins.
Here, the enzyme Thyroid Peroxidase (TPO), in the presence of hydrogen peroxide (H2O2), catalyzes the oxidation of iodide to elemental iodine. This reactive iodine then attaches to tyrosine residues on a large protein called thyroglobulin, a process known as organification or iodination. Depending on whether one or two iodine atoms attach, monoiodotyrosine (MIT) or diiodotyrosine (DIT) are formed. Subsequently, two iodinated tyrosines couple together: a MIT and a DIT combine to form triiodothyronine (T3), the metabolically active hormone, or two DITs combine to form tetraiodothyronine (T4), also known as thyroxine, the prohormone.
T4 and T3 are then stored within the thyroglobulin molecule in the colloid space of the thyroid follicles. Upon stimulation by Thyroid Stimulating Hormone (TSH) from the pituitary gland, thyroglobulin is reabsorbed, cleaved by lysosomal enzymes, and T4 and T3 are released into the bloodstream.
The Double-Edged Sword of Iodine: While absolutely essential, iodine exists on a narrow therapeutic window. Both deficiency and excess can wreak havoc. Chronic iodine deficiency leads to hypothyroidism, goiter, and cognitive impairment. However, excessive iodine intake can also induce thyroid dysfunction, particularly in individuals with pre-existing thyroid conditions or those previously deficient. The Wolff-Chaikoff effect, for instance, describes the temporary inhibition of thyroid hormone synthesis caused by high iodide levels. In susceptible individuals, especially those with autoimmune thyroid disease like Hashimoto's, excessive iodine can exacerbate inflammation and accelerate thyroid destruction, highlighting the need for a balanced approach. This Goldilocks principle – not too little, not too much – is paramount for iodine.
Part 2: Selenium – The Protector and Activator
If Iodine is the architect, meticulously building the raw materials of thyroid hormones, then Selenium is the indispensable master craftsman and vigilant guardian. Often overshadowed, this trace element is a quiet powerhouse, a critical cofactor for a family of enzymes that protect the thyroid from oxidative damage and activate its primary product.
The Trace Element's Odyssey: Selenium, atomic number 34, was discovered in 1817 by Jöns Jacob Berzelius, a Swedish chemist. For many years, it was primarily recognized for its toxicity at high doses, leading to a delayed appreciation of its essentiality. It wasn't until the mid-20th century that scientists began to uncover its crucial biological roles, particularly its integration into specific proteins as selenocysteine, often referred to as the "21st amino acid."
The Selenoproteins: Nature's Master Regulators: The true brilliance of selenium lies in its incorporation into approximately 25-30 human proteins, known as selenoproteins. These are not merely selenium-containing proteins; the selenium atom (as selenocysteine) is an integral, catalytically active component, making them highly efficient enzymes with diverse functions. Among these, several families are absolutely critical for thyroid health and function:
Deiodinases (DIOs): The Activators: This is arguably selenium's most crucial role in thyroid metabolism. The thyroid gland primarily produces T4, which is largely a prohormone. It is T3 that is the biologically active form, responsible for regulating metabolism, growth, and development in target tissues. The conversion of T4 to T3 is carried out by a family of selenium-dependent enzymes called deiodinases.
- Type 1 Deiodinase (DIO1): Found predominantly in the liver, kidney, and thyroid, DIO1 is involved in both the activation of T4 to T3 and the inactivation of T4 and T3 to reverse T3 (rT3) and T2, respectively. It acts as a "scavenger" of thyroid hormones, regulating circulating levels.
- Type 2 Deiodinase (DIO2): Present in the brain, pituitary gland, skeletal muscle, and brown adipose tissue, DIO2 is the primary enzyme responsible for local T4 to T3 conversion, particularly crucial for ensuring adequate T3 supply to the brain, even during states of mild hypothyroidism. This enzyme is exquisitely sensitive to T4 levels, upregulating its activity when T4 is low to maintain local T3 concentrations.
- Type 3 Deiodinase (DIO3): Primarily found in the brain, placenta, and fetal tissues, DIO3 inactivates T4 to rT3 and T3 to T2. Its main role is to protect tissues from excessive thyroid hormone exposure, particularly important during fetal development where precise thyroid hormone levels are crucial.
- Without adequate selenium, the deiodinase enzymes cannot function optimally, leading to impaired T4 to T3 conversion, resulting in lower active T3 levels even if T4 production is sufficient. This is a common scenario in subclinical hypothyroidism and can manifest as persistent hypothyroid symptoms despite seemingly "normal" TSH and T4 levels.
Glutathione Peroxidases (GPx): The Protectors: The synthesis of thyroid hormones within the thyroid gland is a highly oxidative process. The enzyme TPO, as mentioned, uses hydrogen peroxide (H2O2) to oxidize iodide. While essential for iodination, H2O2 is a reactive oxygen species (ROS) that can cause significant oxidative damage to cellular components if not tightly controlled.
- The glutathione peroxidase family of selenoproteins (GPx1, GPx2, GPx3, GPx4) are powerful antioxidant enzymes. They catalyze the reduction of hydrogen peroxide and lipid hydroperoxides to water and harmless alcohols, respectively, using glutathione as a substrate.
- In the thyroid gland, GPx enzymes act as the critical defense mechanism against the oxidative stress generated during hormone synthesis. They prevent damage to thyroid follicular cells, protecting the very machinery that produces thyroid hormones. A deficiency in selenium thus leaves the thyroid vulnerable to oxidative insult, potentially contributing to inflammation and dysfunction.
Thioredoxin Reductases (TrxR): Redox Regulators: The TrxR family (TrxR1, TrxR2, TrxR3) are key enzymes in the thioredoxin system, a central regulator of cellular redox status, cell growth, and protection against oxidative stress. They reduce thioredoxin, which in turn reduces disulfide bonds in other proteins, thereby maintaining protein function and signaling pathways. TrxRs are also critical for regenerating other antioxidants.
Selenoprotein P (SELP): Transport and Antioxidant: SELP is a unique selenoprotein primarily synthesized in the liver. It contains multiple selenocysteine residues and serves as the main transport protein for selenium in the plasma, delivering it to various tissues, including the thyroid. It also possesses antioxidant properties, particularly against peroxynitrite, another potent ROS.
Part 3: The Intertwined Destiny – A Symphony of Synergy
The individual roles of Iodine and Selenium are impressive, but it is their exquisite synergy that truly elevates their importance. They are not merely co-existing; they are co-dependent, each indispensable for the other to perform its function optimally and safely. This is where the story of the duo truly unfolds.
Iodine's Call, Selenium's Response: Imagine Iodine diligently building the framework of a magnificent house (T4). It's a solid structure, but uninhabitable. It sits there, waiting. Selenium, through its deiodinase enzymes, then comes in, transforms the T4 into the active T3, furnishes the house, and makes it ready for living. Without Selenium, Iodine's architectural marvel remains largely inert, a prohormone unable to exert its full metabolic command. This explains why individuals can have "normal" TSH and T4 levels but still experience hypothyroid symptoms if their T4-to-T3 conversion is compromised by selenium deficiency.
The Peroxide Paradox and Selenium's Protective Shield: As discussed, thyroid hormone synthesis inherently generates hydrogen peroxide. This "peroxide paradox" is a masterful example of biological risk management. H2O2 is absolutely essential for the iodination process catalyzed by TPO, yet it is also a highly reactive molecule capable of damaging DNA, proteins, and lipids within the thyroid cells. This is where Selenium's protective role through the glutathione peroxidase enzymes becomes paramount.
When Iodine is abundantly available and the thyroid is actively synthesizing hormones, the production of H2O2 significantly increases. If Selenium levels are inadequate, the thyroid's antioxidant defense system is weakened. The excess H2O2, instead of being neutralized by GPx, can cause oxidative stress, leading to cellular damage and inflammation within the thyroid gland. This is not just a theoretical concern; it has profound implications for thyroid autoimmunity.
The Delicate Balance in Autoimmunity: The Hashimoto's Connection: This synergistic dance takes on a critical dimension in the context of autoimmune thyroid diseases, particularly Hashimoto's thyroiditis, the leading cause of hypothyroidism worldwide. Hashimoto's is characterized by the immune system mistakenly attacking the thyroid gland, leading to chronic inflammation and eventual destruction of thyroid tissue.
A prevailing hypothesis suggests that in individuals genetically predisposed to Hashimoto's, an imbalance between iodine and selenium can act as a trigger or an exacerbating factor. When a selenium-deficient individual consumes sufficient or even slightly supra-physiological amounts of iodine, the increased thyroid hormone synthesis leads to a surge in H2O2 production. Without adequate selenium-dependent GPx enzymes to neutralize this oxidative burst, the thyroid cells become stressed and damaged. This damage can expose cryptic antigens, leading to increased autoantibody production (e.g., anti-TPO antibodies and anti-TG antibodies) and a heightened autoimmune response.
Numerous clinical studies have demonstrated that selenium supplementation (typically 200 mcg/day of selenomethionine) in patients with Hashimoto's can significantly reduce anti-TPO antibody titers, decrease thyroid inflammation, and improve overall thyroid function. This protective effect underscores selenium's role in modulating the immune response and safeguarding the thyroid gland against the very oxidative processes it requires for hormone production. It's a compelling argument for ensuring optimal selenium status before or concurrently with iodine supplementation, especially in those with autoimmune predispositions.
The Thyroid-Brain-Metabolism Axis: Beyond the Gland: The implications of this duo's synergy extend far beyond the thyroid gland itself. Thyroid hormones, particularly T3, are fundamental regulators of virtually every cell and system in the body.
- Brain Development and Function: Adequate T3 is crucial for brain development in fetuses and infants, influencing neuronal migration, myelination, and synapse formation. In adults, T3 impacts cognitive function, mood, memory, and concentration. Impaired T4 to T3 conversion due to selenium deficiency can contribute to "brain fog" and other neurological symptoms associated with hypothyroidism, even with normal T4 levels.
- Metabolic Rate and Energy: T3 is the primary driver of basal metabolic rate. It influences glucose metabolism, lipid synthesis and breakdown, and energy expenditure. A well-functioning thyroid, supported by both iodine and selenium, ensures optimal energy production and utilization, impacting weight management, body temperature regulation, and overall vitality.
- Cardiovascular Health: Thyroid hormones play a role in heart rate, contractility, and blood pressure. Dysregulation can contribute to arrhythmias, hypertension, and hyperlipidemia.
- Reproductive Health: Thyroid dysfunction, both hypo- and hyperthyroidism, can impair fertility, menstrual regularity, and pregnancy outcomes.
- Immune System Modulation: Beyond direct thyroid protection, selenium is a known immunomodulator, influencing the function of various immune cells and contributing to a balanced immune response throughout the body.
Environmental Factors and Modern Diets: The story of iodine and selenium is also shaped by our environment and dietary practices. Iodine deficiency, once a global scourge, has been largely addressed in many developed nations through universal salt iodization programs. However, regions with low soil iodine and populations avoiding iodized salt (e.g., due to processed food consumption, preference for sea salt or specialty salts) remain at risk.
Selenium levels, on the other hand, are highly dependent on soil concentrations, which vary widely geographically. Regions like parts of Europe and China have historically low selenium soils, leading to widespread deficiency. Even in areas with adequate soil selenium, modern agricultural practices, food processing, and dietary shifts (e.g., reduced consumption of organ meats and seafood) can contribute to suboptimal intake. Vegetarian and vegan diets, if not carefully planned, can also be low in selenium.
Part 4: Navigating the Nutritional Landscape – Challenges and Solutions
Given the critical and intertwined roles of Iodine and Selenium, how do individuals ensure optimal status and avoid the pitfalls of imbalance? This requires a thoughtful, informed approach, ideally guided by healthcare professionals.
Assessment of Status: Diagnosing deficiencies or imbalances isn't always straightforward.
- Iodine: Urinary iodine excretion is the most common method for assessing recent iodine intake at a population level. For individuals, 24-hour urine iodine can provide a snapshot, but it fluctuates daily. Blood iodine levels are not routinely used. Often, thyroid function tests (TSH, free T4, free T3) and clinical symptoms are the primary indicators, though they are indirect.
- Selenium: Serum or plasma selenium levels reflect recent intake and body stores. Erythrocyte selenium can indicate longer-term status. Measuring specific selenoproteins like GPx activity or Selenoprotein P can also provide insights into functional selenium status.
- The Full Thyroid Panel: For a comprehensive picture, a full thyroid panel (TSH, free T4, free T3, reverse T3, and thyroid antibodies like TPOAb and TgAb) is essential, especially when symptoms suggest thyroid dysfunction.
Dietary Sources: Prioritizing nutrient-dense whole foods is always the first line of defense.
- Iodine:
- Iodized Salt: The most common and effective source in many countries.
- Seafood: Cod, shrimp, tuna, and other fish are excellent sources.
- Sea Vegetables: Nori, kelp, dulse (caution: kelp can have very high and variable iodine content, risking excess).
- Dairy Products: Milk, yogurt, cheese (due to iodine in animal feed and sanitizers).
- Eggs: A good source, particularly the yolk.
- Selenium:
- Brazil Nuts: By far the richest dietary source; just 1-2 nuts can provide the daily recommended intake. However, selenium content can vary significantly based on soil. Caution against excessive intake due to toxicity risk.
- Seafood: Tuna, cod, sardines, shrimp, salmon.
- Organ Meats: Liver, kidney.
- Meat and Poultry: Beef, turkey, chicken.
- Eggs: Another good source.
- Whole Grains and Legumes: Selenium content is highly dependent on soil levels where they were grown.
- Iodine Supplementation: This is where the story of synergy becomes critical. Never supplement iodine without first ensuring adequate selenium status, especially if you have an autoimmune thyroid condition like Hashimoto's. Starting iodine supplementation in a selenium-deficient individual can exacerbate oxidative stress, worsen thyroid inflammation, and potentially trigger or worsen autoimmunity. Dosing should be conservative, starting low and increasing gradually under medical supervision. The average recommended daily allowance for adults is 150 mcg. Higher doses should be approached with extreme caution.
- Selenium Supplementation: Selenium is generally safer to supplement than iodine, but moderation is key. A common dose studied for thyroid health is 200 mcg per day of selenomethionine, which is well-tolerated and effectively raises selenium status. Forms like sodium selenite are also available but may have different bioavailability. Toxicity can occur at very high doses (e.g., consistently above 800 mcg/day), leading to symptoms like hair loss, brittle nails, and neurological issues.
- The Broader Micronutrient Context: It's important to remember that iodine and selenium don't operate in a vacuum. Optimal thyroid function also relies on other essential micronutrients:
- Zinc: Involved in TSH production and thyroid hormone action.
- Iron: Essential for TPO enzyme activity.
- Vitamin D: Modulates immune function and may play a role in autoimmune thyroid disease.
- B Vitamins: Crucial for energy metabolism and overall cellular function.
A holistic approach that considers the entire nutritional landscape, alongside lifestyle factors (stress management, sleep, physical activity), provides the most robust foundation for thyroid and metabolic health.
Conclusion
The story of Selenium and Iodine is a compelling testament to the exquisite intelligence of the human body and the intricate dance of micronutrients. They are not merely individual elements to be consumed, but an indispensable duo, each playing a distinct yet interconnected role in the grand symphony of thyroid hormone production, activation, and protection. Iodine lays the foundation; Selenium activates the finished product and defends the workshop from the very processes required for its creation.
For the knowledgeable audience, understanding this synergy moves beyond simple dietary recommendations. It illuminates the profound biological implications of imbalance and highlights the critical need for a nuanced approach to nutrition and supplementation. In a world where thyroid disorders are increasingly prevalent, recognizing the essential partnership of Iodine and Selenium is not just academic; it is a vital step towards unlocking optimal thyroid function, boosting metabolism, and fostering a healthier, more vibrant life. Their dance is a silent, constant reminder of the profound wisdom embedded in nature's smallest details, urging us to listen, learn, and nourish our bodies with the respect they deserve.